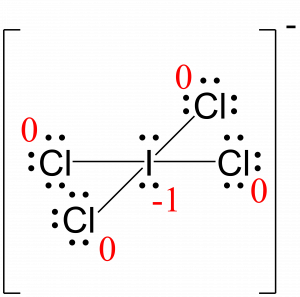 
Look at the image above at the SCN- ion to the left and the way they calculated formal charge:įor the sulfur atom, they took the number of valence electrons (6) and subtracted it by the total number of lone electrons (2) and single bonds with carbon (3 ). This might may not be the most technical but it's the easiest way to remember it! 
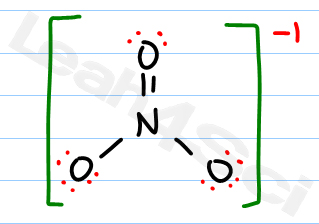
The easiest way to calculate the formal charge of an atom is to do (# of valence electrons - # of dots - # of dashes). The -1 charge indicates that there is one more electron, so there must be a total of 24 valence electrons represented in the LDS.ĭraw the central atom, which in this case, is nitrogen since there is only one nitrogen atom.ĭraw the 3 oxygen atoms surrounding nitrogen and three single bonds connecting them to nitrogen. But.NO3- is a polyatomic ion and there is a charge attached to the molecule as a whole. Since there are three oxygen atoms, we must account for each: 5+6+6+6 = 23. Recall some steps for this process that we discussed in the last study guide:Ĭount the number of valence electrons that the molecule has in total. Let's try drawing the lewis dot structure (LDS) of the polyatomic ion NO3. How do you know when a structure has resonance? This will make more sense as we take a look at several examples. However, resonance is all about representing a molecule in varying ways just to make it clear that the molecule is actually an average of those representations in space. A common misconception is that structures that have resonance are truly able to exist with different bond connections.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
